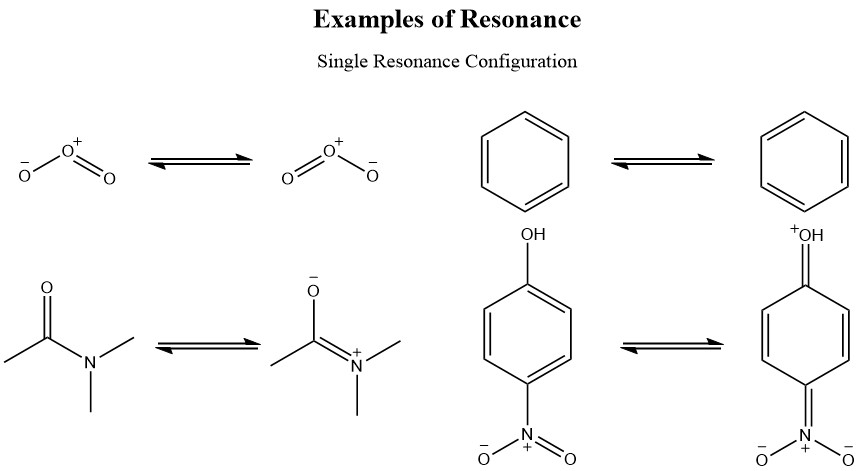
Get Benzene Resonance Structures Gif. Due to resonating structures or delocalized pi. Modern descriptions of the benzene structure combine resonance theory with molecular orbital theory.
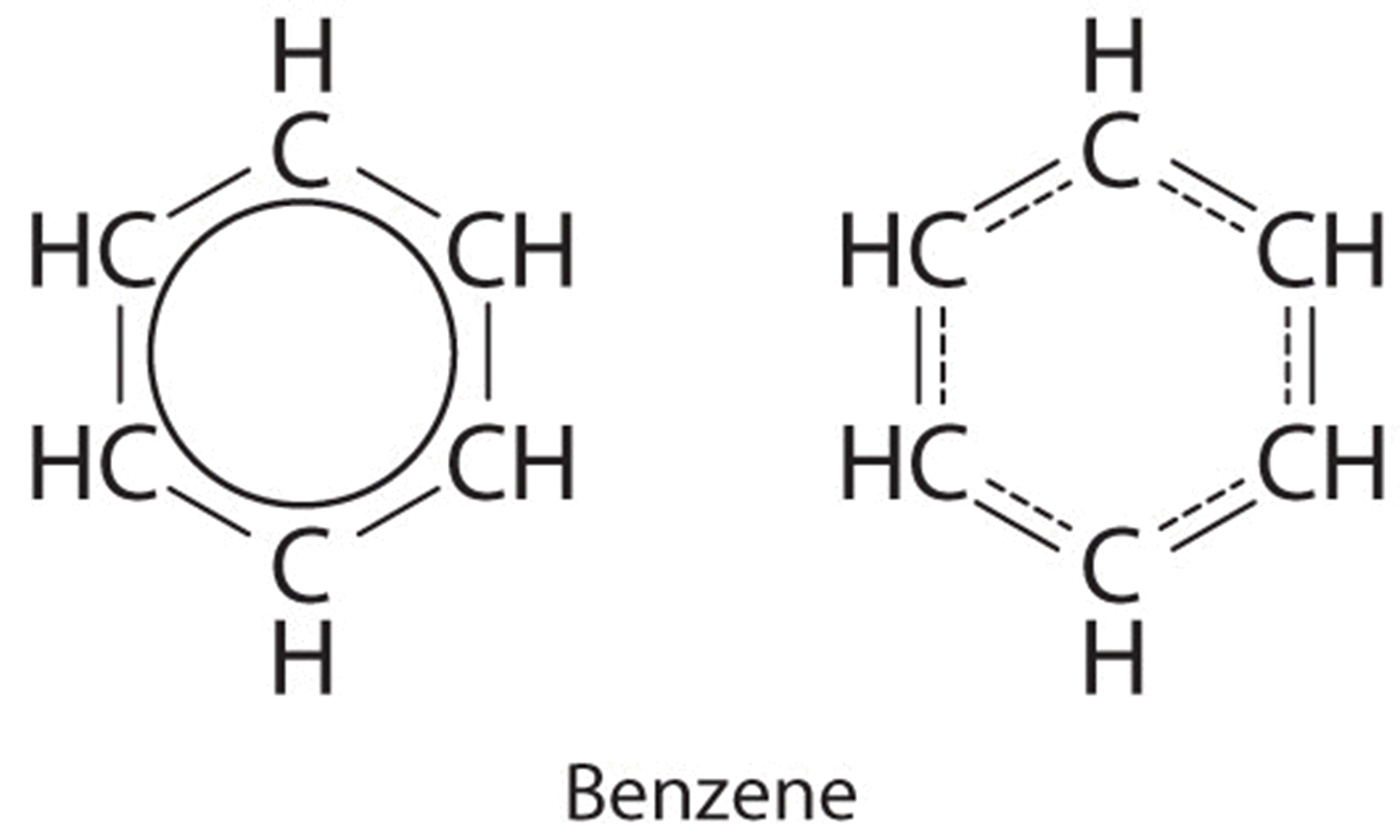
So benzene undergoes substitution reactions.
A description of the kekulé structure for benzene and the reasons (including hydrogenation kekulé was the first to suggest a sensible structure for benzene. The pi molecular orbitals of benzene, and how to build up the mo diagram; According to the resonance model, benzene does not exist as a pair of rapidly interconverting conjugated trienes but has a single structure that cannot be represented by formulations with. As such, benzene readily crosses the alveolar membranes and is taken up by circulating blood in pulmonary vessels.