Get Benzene Resonance Structures Lewis Background. It highlights and contrasts the lewis structures which represent a particular molecule. Resonance structures are sets of lewis structures that describe the delocalization of electrons in a polyatomic ion or a molecule.
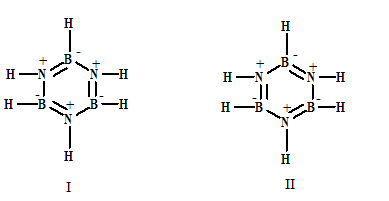
Benzene has 2 resonance structures but taken individually none show the delocalisation of electrons and they the resonance hybrid is the approximate intermediate of the contributing structures, but the this catalyst is usually a lewis acid (alcl3, febr3…) or in the case of the nitration of benzene.
How to draw the lewis structure for benzene (c₆h₆)? Where there are double bonds, there could actually be single bonds and vice versa. Molecular formula and molecular geometry. Modern descriptions of the benzene structure combine resonance theory with molecular orbital theory.
