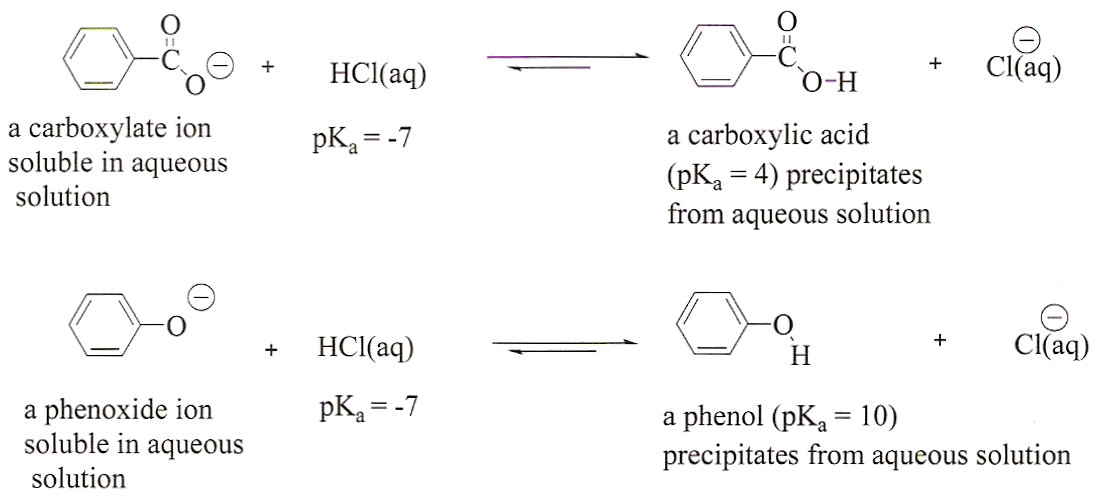
View Benzoic Acid And Naoh Equation Background. I choose to divide the problem into two parts; Show the line structure of benzoic.
Naoh will react more with hcl than ethyl benzoate.
Write the balanced equation between benzoic acid and naoh. The acidic portion of benzoic acid is the carboxyl group, and it reacts with a base to form a salt. How many ml of 0.10 m naoh are required to reach the equivalence point in the titration of 25.00 ml of a 0.070 m weak acid solution. Examples of arrhenius acids include hydrochloric acid, hcl, sulfuric acid, h2so4, and acetic acid (vinegar), ch3cooh.
